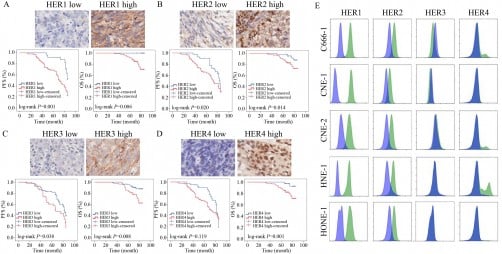
FDA Restricts COVID-19 Vaccine Access for Healthy Individuals
Federal officials in the United States have confirmed that COVID-19 vaccines remain available to the public, despite new restrictions that limit their approval. On Wednesday, the U.S. Food and Drug Administration (FDA) authorized updated COVID-19 vaccines primarily for seniors and younger individuals with underlying health conditions, narrowing the previous eligibility that included all individuals aged six months and older.
Dr. Marty Makary, the FDA Commissioner, asserted on social media that “100% of adults in this country can still get the vaccine if they choose. We are not limiting availability to anyone.” Alongside this, Karoline Leavitt, White House press secretary, reiterated that the FDA’s decision does not affect the availability of COVID-19 vaccines for Americans who wish to receive them, emphasizing the importance of individual choice.
Despite these statements, experts argue that healthy individuals who do not meet the specified criteria may face significant barriers to access. For these individuals, obtaining an updated COVID-19 vaccine will likely require an “off-label” prescription, a practice that involves using medications outside their FDA-approved guidelines.
Dr. Jake Scott, an infectious disease specialist at Stanford Health, highlighted the discrepancy between theoretical availability and practical accessibility. He stated, “Technical availability and practical accessibility are very different things.” The policy shift has replaced straightforward access at pharmacies with a system requiring consultations with healthcare providers, navigating insurance complexities, and finding pharmacies willing to administer the vaccine.
Historically, most COVID-19 vaccinations have occurred in pharmacies, but off-label prescribing necessitates a visit to a physician. Experts indicate that some healthcare providers may be hesitant to prescribe off-label, and pharmacies might adopt a more conservative stance regarding vaccine distribution. Dr. William Schaffner, an infectious disease expert at Vanderbilt University, noted that the current climate of uncertainty may discourage pharmacies from risk-taking in administering vaccines.
Additionally, Dr. Jerome Adams, who served as U.S. surgeon general during the Trump administration, pointed out that approximately 11% of U.S. adults are uninsured and lack access to a doctor. He criticized the FDA’s assurances, stating, “To be frank and objective, unless he’s also giving everyone access to free healthcare, he seems to be reneging on his promise.”
Insurance coverage for off-label prescriptions can vary, potentially imposing substantial out-of-pocket costs for a vaccine that has traditionally been offered at no charge. Dr. Kelly Moore, president and CEO of immunize.org, emphasized that relying on off-label prescriptions may benefit only a limited number of individuals who have both the financial means and access to cooperative physicians.
Claims of universal access to COVID-19 vaccinations have also been challenged. According to Dr. Scott, there are currently no options available for children under five who do not have underlying conditions that elevate their risk of severe illness. Emergency use authorizations for COVID-19 vaccines have been rescinded, as stated by Robert F. Kennedy Jr., U.S. Department of Health and Human Services Secretary, which means that Pfizer’s COVID-19 vaccine, Comirnaty, is no longer authorized for children younger than five. In contrast, Moderna’s Spikevax vaccine is available for children as young as six months, but only if they have a qualifying health condition.
As uncertainty looms, the independent advisers to the U.S. Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) have yet to provide guidance on the updated COVID-19 vaccines. Their forthcoming decisions could significantly influence vaccine access across various states. Dr. Georges Benjamin, executive director of the American Public Health Association, addressed the potential for confusion at the service level, noting that some pharmacists and nurse practitioners may not be permitted to administer vaccines not listed on the CDC’s schedule.
Consequently, states may be faced with varying regulations, leading to 50 different interpretations of how vaccines can be administered. Some states may require legislative changes to adapt their policies, which could delay access until January.
As the U.S. navigates these complexities, experts remain concerned about the future of COVID-19 vaccination efforts. Dr. Schaffner remarked on the inevitability of ongoing COVID-19 challenges, stating, “We anticipate with full confidence, I’m afraid, that there will be a substantial winter increase in COVID and that we will have, as a consequence, hospitalizations, people admitted to intensive care units and some deaths.”
The evolving landscape of COVID-19 vaccine access underscores the necessity for clear communication and effective solutions to ensure that all individuals, regardless of health status, have the opportunity to protect themselves against the virus.